Category: Future Physicians, Health Magazine
Title:Old Drug, New Promise in Clinical Trial for Parkinson’s
Date Published: April 15, 2016

In a small scale study, Georgetown University Medical Center researchers have discovered that a drug used to treat leukemia improves the symptoms of Parkinson’s disease.
In the proof of concept study, 12 patients received nilotinib, an FDA-approved drug for leukemia. The drug improved cognition, motor skills, and non-motor function in patients with Parkinson’s disease and Lewy body dementia. In addition, it led to statistically significant and encouraging changes in toxic proteins linked to disease progression.
Charbel Moussa, director of Georgetown’s Laboratory of Dementia and Parkinsonism, and Fernando Pagan, director of Georgetown’s Movement Disorders Program, led the trial, which was supported by the Georgetown-Howard Universities Center for Clinical and Translational Science. Preliminary data were presented at Neuroscience 2015, the annual meeting of the Society for Neuroscience, in October. (Update: Final data from this study was published July 11, 2016 in the Jouranl of Parkinson’s Disease)
Moussa conducted the preclinical research that led to the discovery of nilotinib for the treatment of neuro-degenerative diseases, then partnered with Pagan for the clinical study. “To my knowledge, this study represents the first time a therapy appears to reverse—to a greater or lesser degree depending on stage of disease—cognitive and motor decline in patients with these neurodegenerative disorders,” says Pagan.
Investigators report that the six-month study of nilotinib, a treatment for chronic myelogenous leukemia or CML, produced benefit for all study patients who completed the trial (11 of 12), with 11 patients reporting meaningful clinical improvements. The study’s primary objective was to test safety. Researchers say that use of nilotinib, in doses much smaller than those used to treat cancer, was welltolerated with no serious side effects.
Moussa and Pagan found that the drug penetrates the blood-brain barrier in amounts greater than dopamine drugs, but the observed efficacy in cognition, motor skills, and nonmotor function improvement for many patients was the most dramatic result. The investigators report that one individual confined to a wheelchair was able to walk again; three others who could not talk were able to hold conversations.
Alan Hoffman, a retired professor, was diagnosed with Parkinson’s disease in 1997 and has participated in several clinical trials with no benefit, he says, until he enrolled in Pagan’s study. Before nilotinib, he was unable to do simple household tasks, but after the trial he said, “Now, I empty the garbage, unload the dishwasher, load the washer and the dryer, set the table, even take responsibility for grilling.”
In the three weeks prior to enrolling in the study, Hoffman says he fell eight times, but only fell once during six months on the study.
His speech has improved, as has his thinking. “My wife says it’s life-changing for her and for my children and grandchildren,” Hoffman says. “To say that nilotinib has made a change in our lives is a huge understatement.”
While the results of the small trial are promising, they should be interpreted with appropriate circumspection— there was no control group for comparison. Also, nilotinib was not compared with a placebo or other medications used to treat Parkinson’s in the study.
Moussa and other Georgetown researchers are planning larger clinical trials with nilotinib for patients with Parkinson’s and other similar diseases including Alzheimer’s disease, likely to begin this year. Moussa is an inventor on a Georgetown University patent application for use of nilotinib for the treatment of neurodegenerative disease.
Support for the next phase of the Parkinson’s trial includes a recent $1 million gift from the Lasky-Barajas Family Fund to the Nilotinib Clinical Research Fund in the Translational Neurotherapeutics Program at Georgetown University Medical Center. The phase II Alzheimer’s trial also continues to receive funding from individuals and organizations including the Alzheimer’s Drug Discovery Foundation.
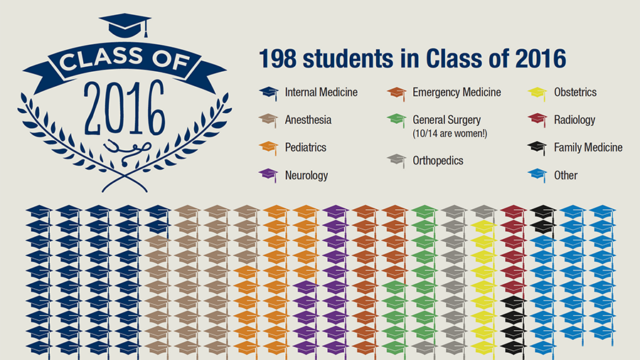
Match Day is a pivotal day in the life of graduating medical students.

Through a series of key building enhancements and accessibility projects, the proposed expansion addresses the urgent age, capacity, and technology issues of the hospital today.
The Huntington Disease Care, Education & Research Center at Georgetown has been designated as a Center of Excellence by the Huntington's Disease Society of America.